Technical File: Secure your technical dossier and CE marking.
A rejected technical file doesn't just cost months of Time-to-Market. Missing data, justification errors, cascading inconsistencies, non-compliance with applicable standards... These flaws put your certificate at risk and cost you enormous time and money.
MAC's AI assistant acts as a ruthless pre-Notified Body to bulletproof your submission and documentation maintenance.
Completeness Verification (Submission Readiness)
Never submit an incomplete file again. Before any interaction with the Notified Body, MAC scans your entire documentation to ensure no document, signature, or mandatory data is missing. Your technical file exits with 'Submission-Ready' status.

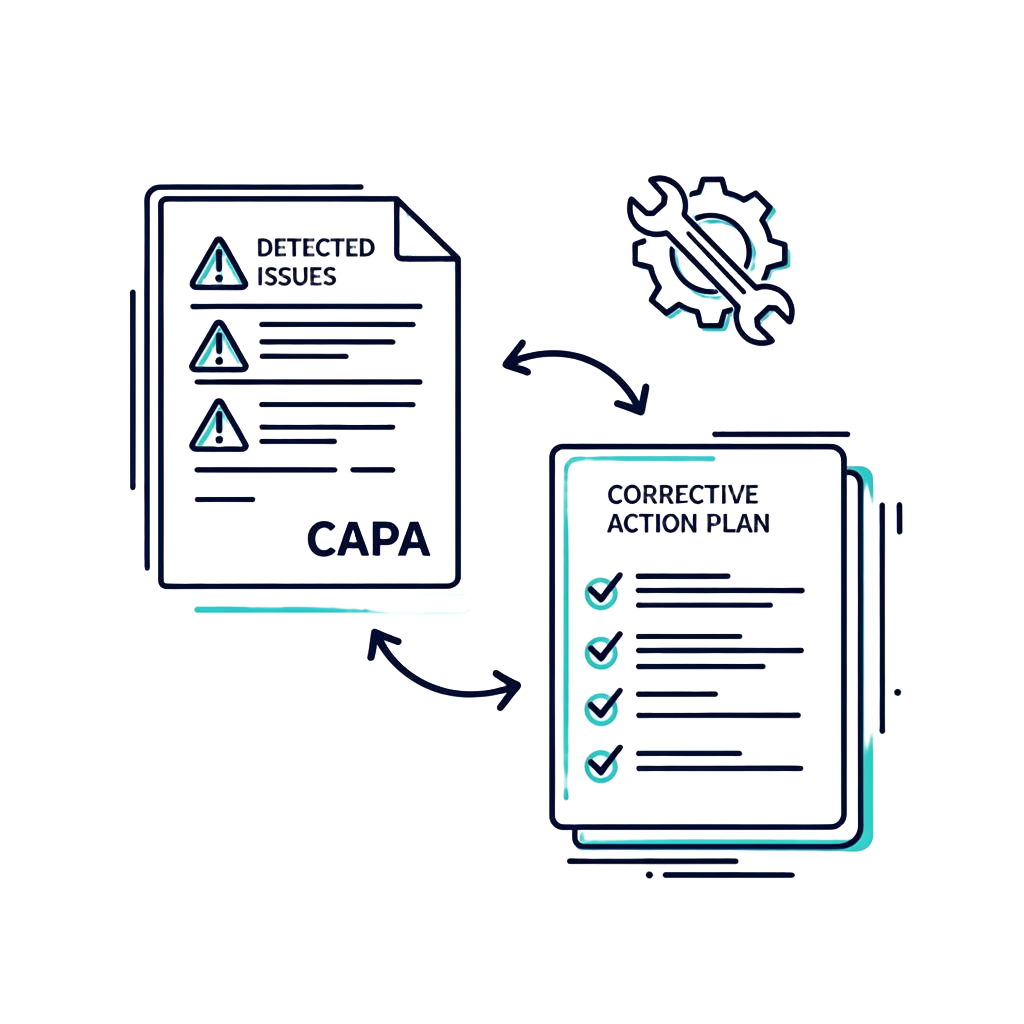
Action Plan Generation for Technical File
For each gap, omission, or inconsistency detected in your dossier architecture, the AI doesn't just raise an alert. It instantly generates a detailed corrective action plan so your engineers can fix issues without wasting time on interpretation.
Consistency & Traceability (GSPR / Risks / Clinical)
The traceability matrix is often manufacturers' Achilles heel. The AI relentlessly tracks inconsistencies and logical breaks between your General Safety and Performance Requirements (GSPR), your risk management file (ISO 14971), and your Clinical Evaluation Report (CER). If an identified risk isn't clinically covered, MAC flags it immediately.

R&D Traceability (Design Inputs / Outputs & V&V)
The development cycle of a medical device constantly evolves, sometimes creating a gap between the initial need and the final product. The assistant automatically verifies the uninterrupted continuity between your Design Inputs, Design Output specifications, and Verification and Validation (V&V) elements.
Compliance with Applicable Requirements & Standards
The state of the art is constantly evolving. MAC detects the applicability of regulatory texts to your specific device and verifies that you have responded precisely (and justified your choices) to the dozens of harmonized standards and regulations applicable to your product.

MDR / IVDR Classification Verification
A classification error can force you to restart your entire clinical strategy. MAC performs rigorous verification of your medical device (MDR) or in vitro (IVDR) device classes according to complex European classification rules.

Ready to see MAC in action?
Connect your document repository and let your AI assistant audit your compliance today.
Roadmap — Vote for the next features
Join the Beta to unlock this feature for free before everyone else
Notified Body Formatting
The assistant automatically structures your technical dossier according to your evaluation body's specific templates and requirements.
Non-Conformity Report Response
Semantic analysis of Notified Body feedback and automatic preparation of a justified, documented response.
Product Change Management
Automatic and instant evaluation of the regulatory impact of a design change on your entire dossier.
Automatic IFU Verification
The AI cross-references your Instructions for Use (IFU) with your risk management file to ensure no warning is missing.