MDR / IVDR Transition: Unlock your products, secure your legacy.
Moving to the new European regulations isn't a simple administrative formality — it's a wall of technical complexity. The MAC assistant produces your status review in 24 hours and delivers a clear roadmap to unlock products currently frozen by the "significant change" rule, and to secure your business for the long term.
Did you know? Your devices may be frozen.
As long as you are not MDR/IVDR-certified, the "significant change" rule applies: any significant modification to your existing product forces you to move immediately to the new regulation. The result? Your legacy products are frozen, your innovation is stalled, and your competitors are moving ahead. You can no longer wait.
Your roadmap to MDR / IVDR compliance
6 key steps where the MAC assistant turns every friction point into a competitive advantage.
The starting diagnostic
The starting diagnostic
Your reality
"We've always done it this way." Your files are dense and you don't know where to start or how much effort will be required.
What MAC brings
Just connect your existing documentation. Within 24 hours flat, MAC runs an exhaustive Gap Analysis of your QMS and Technical Files.

The prioritized roadmap
The prioritized roadmap
Your reality
The workload is colossal. The mountain of regulatory gaps creates a sense of panic and dispersion.
What MAC brings
The AI generates a ready-to-execute action plan (CAPA) and intelligently prioritizes critical workstreams. Your team members know exactly what to do.
Execution under close watch
Execution under close watch
Your reality
By modifying dozens of documents in parallel, you lose overall consistency — the domino effect.
What MAC brings
Throughout your work, the assistant checks in the background that everything stays consistent. The AI alerts you immediately in case of contradiction.

Submission to the Notified Body
Submission to the Notified Body
Your reality
The fear of rejection by the Notified Body (NB), meaning loss of the right to sell and months of lost revenue.
What MAC brings
You submit a unified, "Submission-Ready" Technical File, pre-audited by an AI that applies the same level of rigor as your NB..
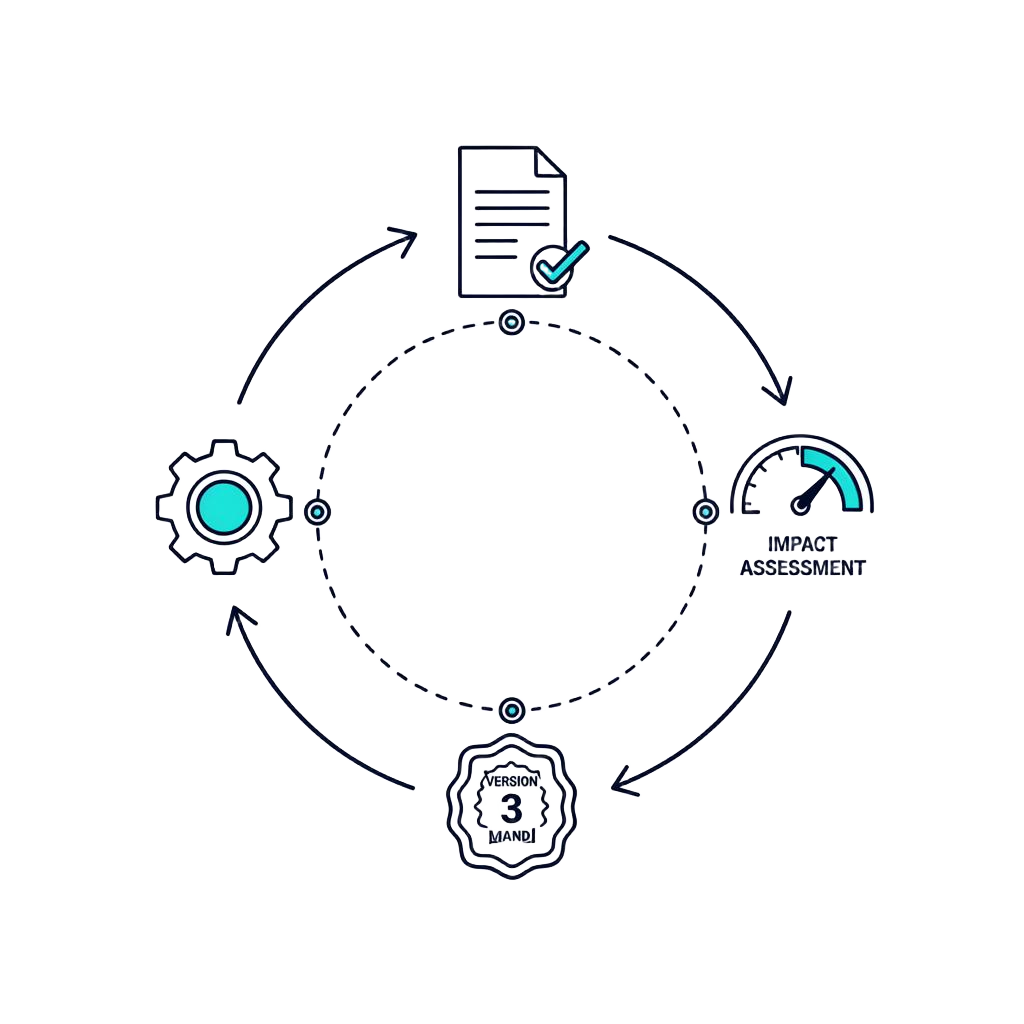
Lifecycle management (Change Control)
Lifecycle management (Change Control)
Your reality
MDR certificate in hand... but R&D already wants to evolve the product. The risk? That a simple change breaks compliance.
What MAC brings
Agility restored. MAC instantly assesses the regulatory impact of every change across your entire documentation.
Continuous audit and automated regulatory monitoring
Continuous audit and automated regulatory monitoring
Your reality
Maintaining compliance is expensive. Audits and regulatory monitoring are a permanent burden for teams under pressure.
What MAC brings
Complete peace of mind. Continuous evaluation replaces tedious audits, and the AI automatically cross-checks new standards against your files.

A controlled transition — an investment that pays for itself.
Don't lose the right to sell your legacy products because of a poorly managed transition. Investing in the MAC assistant is immediately offset by the massive time saved on cross-document analysis, by optimized external consulting budgets, and by the preservation of your product catalog.
100% Plug & Play — compatible with your tools


Stop being at the mercy of the new European regulations.
Take back control of your compliance and give your teams the agility they need to evolve their medical devices.